Karriere bei Sacher

Related Products
Application
Indium SpectroscopyIndium is a very reactive metal of the third group of the table of elements. It was first discovered by Reich and Richter in 1863 using spectroscopic investigations. Due to its typical blue color of the spectral lines it was named after the collor Indigo. It is characterized by one valence electron. The natural abundance of Indium isotopes is 113In(4.3%) and 115In(95.7%).
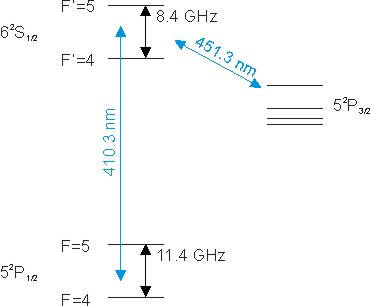
There are several optical transitions at 410nm and 451nm. Their electronic levels are shown in the excerpt of the level scheme. Due to the low natural abundance of 113In, only the transitions of 115In are shown.
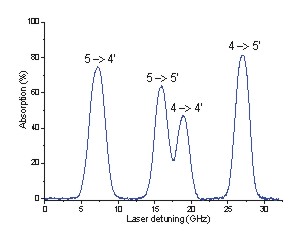
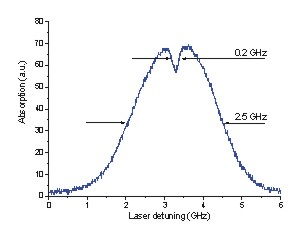
All four possible optical transitions at 410nm are observed during an In absoption experiment with our tunable external cavity diode lasers as shown on the above absorption spectra. The lower figure shows the Lamb dip of one transition, investigated with a typical saturation absorption experiment.
These experiments have been performed with our TEC100 Blue Littrow Laser System, as well as with our TEC500 Blue Littman Laser System. For more details, please check our publications.