Related Products
Application
Absorption Spectroscopy: An IntroductionAbsoption Spectroscopy is a very powerful method for measuring trace gas concentrations over distances or within difficult enviroments. It works best for atoms or small molecules. For large molecules, please check for Raman Spectroscopy.
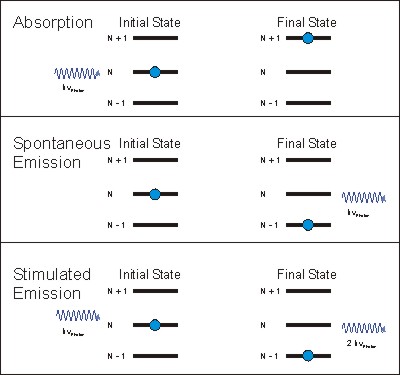
There are three different processes of light interacting with gases: Absorption, Spontaneous Emission and Stimulated Emission. For Absorption Spectroscopy, Absorption and Spontaneous Emission are the relevant processes.
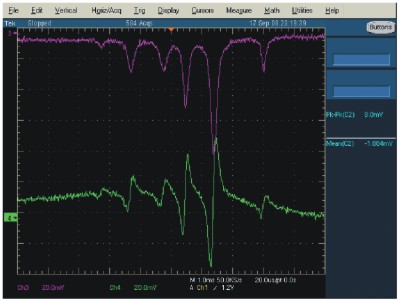
For Absoption Spectroscopy, the laser light passes thru a cavity which is filled by a gas sample for investigation. When tuning the laser emission wavelength over the absorption line of the sample gas, a part of the light intensity is absorbed by the gas and re-emitted after a typical delay time. Since the re-emitted light does not have a direction preference, it is emitted in all space directions. The results is a reduction of the light intesity at the wavelength of the absorption line.
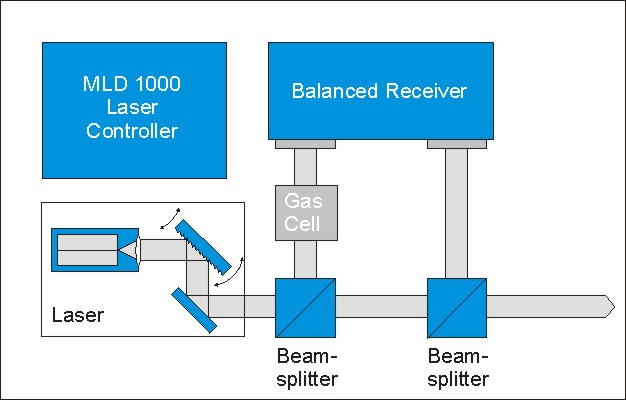
For applying Absorption Spectroscopy, a tunable laser system and a photodetector is required. A common setup for spectroscopy consists of a tunable diode laser system together with a laser controller. The controller allows to tune the emission wavlength of the laser system over the absorption line of the investigated gas. The laser beam is splitted into two beams. One beam is the measuring beam which passes the gas sample. The second beam is a reference beam which reduces the noise level of the experiment. Both laser beams are coupled to a balanced receiver which measures the difference in between and gives the pure absorption value. This method allows measuring gas concentrations into the ppm regime.
Applications Examples are given on this website for various gases or vapours. Please check our Technical Notes and Publications or contact us for more detailed information.

 X500 Manual Tuning
X500 Manual Tuning